Sedimentary rock types
Terrigenous sediments (clastic)
These are land derived sediments sub-divided further on the basis of their grain size.
- Group A – Clays (particles with a diameter less than 0.004 mm)
- Group B – Silts (particle diameter 0.004 to 0.06 mm)
- Group C – Sands (particle diameter 0.06 mm to 2 mm)
- Group D – Rudites (coarser rock fragments).
Clay
1- Clay minerals are hydrous platy aluminosilicates.
2- Clay minerals can be subdivided into five important groups with different chemical and physical characteristics; kaolinite, illites, smectites, chlorites and glauconites.
3- “shale”, generally used to name those argillaceous sediments, mostly describes the tendency of those materials to split, especially when they have been exposed to high compactions and pressures.
4- Gumbo is a term used to describe claystone formations that absorb water, hence, hydrating shales tend to expand. Sticking mechanisms are associated with such formations:
Contraction of the wellbore behind the BHA makes it difficult to trip pipe out. Large clumps of gumbo will fall into the wellbore and will eventually stick to the BHA. Shale inhibitors can be added to the drilling fluid in order to restrict or avoid such phenomenon.
Silts
- Silts are clastic sediments, intermediate in size between clays and fine sands (particle diameter 0.004 to 0.06 mm).
- While sand may be silt free, most clays and claystones contain about 35% silt or more (thus named silty claystones or argillaceous siltstones).
- Very abrasive siltstones can be encountered in the drilling processes and, therefore, frequent bit changes become necessary.
Sands
- Sands, like silts, are defined by their grain size (0.062mm) and not by their mineralogy.
- They are frequently porous and permeable, thereby satisfying three basic requirements for major aquifers and hydrocarbon reservoirs.
Rudites
- grain size exceed 2 mm in diameter.
- Are common in both the carbonate and pyroclastic groups.
Pyroclastic sediments
These are derived by volcanic eruption into the air. .
Carbonates
1- This major group of sediments is fundamentally different to the terrigenous (or siliclastic) and pyroclastic rocks just discussed.
2- The Chemical Components of Carbonates
- Aragonite
- Magnesian
- Calcite
- Dolomite
All these minerals, know as polymorphs of calcium carbonate (CaC03), present different degrees of chemical stability depending on the environmental characteristics of the depositional basin.
3- The physical components of carbonates
- The grain types either mineral or biological.
- The matrix which consists of the fine material.
- The cement which grows in the pore spaces of the sediment after it deposition.
- The pore space remaining after cement has taken place.
4- The Classification of Limestone Rocks Four more terms are frequently used to describe grain size in carbonate rocks.
- Group A – Calcilutite up to 0.004 mm grain diameter
- Group B – Calcisiltite 0.004 to 0.065 mm
- Group C – Calcarenite 0.065 to 2 mm
- Group D – Calcirudite above 2 mm grain diameter
5- Certain specific types of limestones like chalk, marl, bituminous limestones and dolomite can be as well mentioned at this stage.
- Chalk This is a soft white limestone composed of the tests (or skeletons) of once floating micro-organisms.
- Marl This is a calcareous clay – generally an intermediate mixture of terrigenous clay and micrite.
- Bituminous Limestones These are micrites which contain much organic or carbonaceous matter, mostly in the form of tarry hydrocarbons which are usually described as bitumen.
- Dolomite This term is applied to limestones where the calcium carbonate has been completely replaced by the mineral dolomite.
Evaporites
These sediments, which include mineral salts such as anhydrite. gypsum and rock salt (halite evaporation). They play an important role in petroleum geology, being excellent cap rocks for oil or gas reservoirs.
- They are also very plastic and thick salt sequences deform and flow to produce salt domes.
- Salt movements frequently produce hydrocarbon traps.

Carbonaceous rocks
1- Coal
2- Oil Shale
This is more an economic rather than geological term, and refers to argillaceous sediments with an organic content of at least 5%, but generally meaning considerably higher (20 to 50%). They must be sufficiently rich in organic matter to yield free oil on heating. They generally form in lakes where algae matter decays in a strongly reducing (or anaerobic) environment, thereby preserving the organic material. This is referred to as “sapropelic” matter and is a good source for oil. Marine equivalents are also known.
3- Bituminous Limestone
This is again an economic rather than geological concept being the direct carbonate equivalent of terrigenous oil shales. Such sediments may form in lagoons behind a reef.
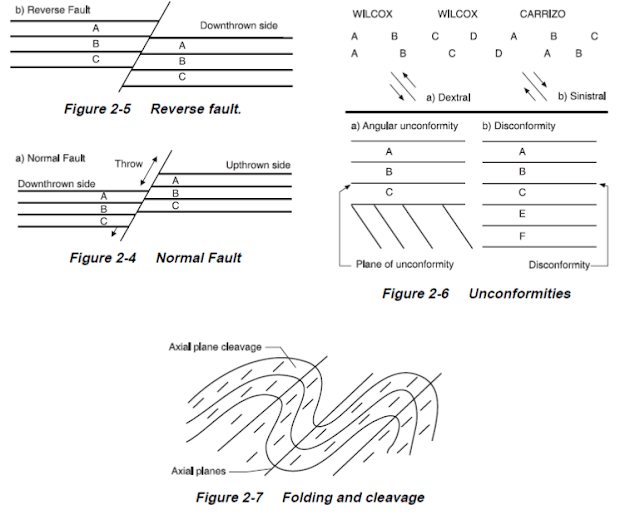
Structural Geology
Abnormal Pressure
Reservoirs that do not have a connection with the surface are totally surrounded by impermeable formations. In such cases, the overlying rock formations do have a bearing on reservoir pressure. What happens in this case is that the heavy weight of the overlying beds presses down and squeezes the reservoir. Since the water in the reservoir cannot escape to the surface, the reservoir pressure builds up to abnormally high amounts.
Another way in which abnormally high pressure can exist is when there is an artesian effect (Figure 2-13). In this case, the reservoir does connect with the surface. However, the outcrop to the surface is on the side of a hill or mountain, at an elevation much higher than the part of the reservoir buried below the level plain. A well drilled at this point spouts water like a fountain. The water tries to seek its own level. Such wells are called artesian wells.